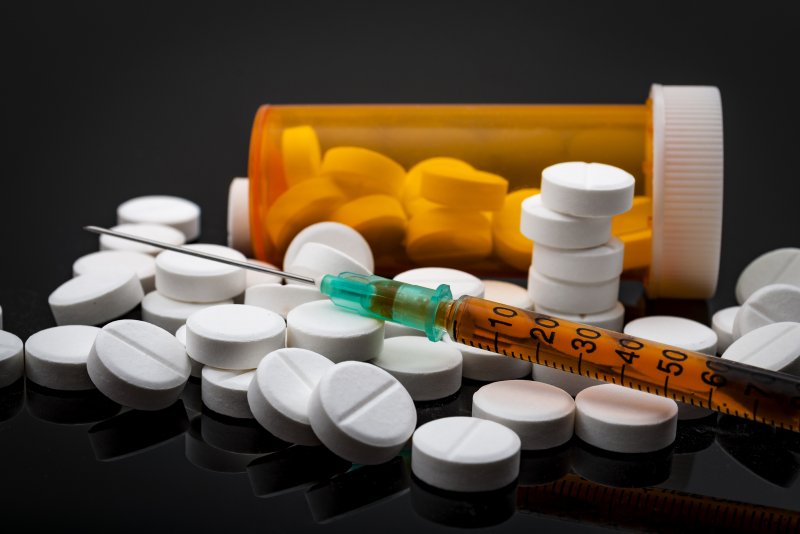
The first generic naloxone nasal spray to treat opioid overdose has received approval from the U.S. Food and Drug Administration.
Teva Pharmaceuticals' lifesaving product is also the first generic naloxone nasal spray approved for use by people without medical training. There was already a brand-name spray, Narcan, for emergency use by untrained people, such as family members and bystanders.
The need is urgent. On average, more than 130 Americans die every day from overdoses of opioids -- including prescription painkillers such as fentanyl, oxycodone [OxyContin], hydrocodone (Vicodin) and morphine, as well as illegal drugs such as heroin or drugs sold as heroin, the FDA said.



 The U.S. Department of Agriculture confirmed the presence of New World screwworm in a 3-week-old calf...
The U.S. Department of Agriculture confirmed the presence of New World screwworm in a 3-week-old calf... Claude Lemieux’s brain is being donated to the Boston University CTE Center to research the long-term...
Claude Lemieux’s brain is being donated to the Boston University CTE Center to research the long-term... A smart drug that stops cancer cells “hiding” from treatment can shrink tumours by at least...
A smart drug that stops cancer cells “hiding” from treatment can shrink tumours by at least...